RESEARCH ARTICLE
The Vest™ High-Frequency Chest Wall Oscillation System Compared with Manual Chest Wall Physiotherapy for Managing Airway Clearance in Patients with Complex Neurological Disorders: A UK-based Cost-Effectiveness Analysis
Mehdi Javanbakht1, *, Atefeh Mashayekhi1, Mohammad Montazeri2, Mohsen Rezai Hemami3, Michael Branagan-Harris4
Article Information
Identifiers and Pagination:
Year: 2019Volume: 7
First Page: 1
Last Page: 8
Publisher Id: TOPHARMEJ-7-1
DOI: 10.2174/1874129001907010001
Article History:
Received Date: 25/02/2019Revision Received Date: 14/05/2019
Acceptance Date: 16/05/2019
Electronic publication date: 31/05/2019
Collection year: 2019
open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: https://creativecommons.org/licenses/by/4.0/legalcode. This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:
Weakness in the muscles used to breathe and swallow can lead to difficulties with airway clearance for people with neurological disorders including neuromuscular disease and cerebral palsy. This can lead to aspiration and other respiratory problems which are the leading causes of death in patients with complex neurological disorders. The Vest™ system supports airway clearance through the use of High-Frequency Chest Wall Oscillation (HFCWO) to loosen secretions in the chest by reducing their viscosity.
Objective:
To assess the cost-effectiveness of the Vest™ system versus Manual Chest Wall Physiotherapy (MCWP) for airway clearance in patients with neurological disorders including neuromuscular disease and cerebral palsy.
Methods:
A decision-analytic Markov model was developed to estimate the cost-effectiveness of HFCWO and MCWP over 5- and 10-year time horizons. Costs were estimated from the perspective of the UK National Health Service and personal social services. The main input parameters in the model were: rates of respiratory infection, respiratory-related hospitalisation, antibiotic use for respiratory infection and cost of the Vest™ system. The input parameters were informed by existing clinical guidelines and literature. Sensitivity analyses were conducted to explore uncertainties around the input parameters.
Results:
Over 5- and 10-year time horizons, the Vest™ system results in more quality-adjusted life-years and lower costs per patient. The Vest™ system has a high probability of being cost-effective (>98%) at willingness-to-pay thresholds of £20,000 and £30,000. The estimated average cost saving per patient over a 5-year time horizon is £5,660 (SD = £2362). Our results show that the Vest™ system may result in approximately £6 million cost savings per 1,000 patients with complex neurological disorders in 5 years. Per 1,000 patients, 2,442 hospital admissions and 49,868 bed days could be averted with the use of the Vest™ system. Our results were generally robust to the sensitivity analyses performed.
Conclusion:
The Vest™ system results in fewer respiratory infection episodes and hospitalisations, and thus less National Health Service (NHS) resource use, than current practice. Therefore, it is highly likely to be a cost-saving strategy.
1. INTRODUCTION
Weaknesses in the muscles used to breathe and swallow can contribute to respiratory problems in patients with complex neurological disorders including neuromuscular disease and cerebral palsy [1]. In particular, these patients may have difficulty in coughing and clearing their airway effectively. Ineffective airway clearance can lead to aspiration of saliva, liquids, or food. These patients often present with conditions such as pneumonia or other lower respiratory tract infections, pleural effusion, or respiratory failure resulting from ineffective airway clearance. These conditions often require lengthy hospital admissions and significant resource use [2]. Respiratory problems are the main cause of death in people with complex neurological disorders [2].
UK National Health Service (NHS) Hospital Episode Statistics (HES) data for 2016/17 show that nearly 19,000 non-elective emergency admissions for patients with neuromuscular disease were related to a respiratory condition. There were also 4,400 non-elective emergency respiratory-related admissions for patients with cerebral palsy. These admissions resulted in significant use of NHS resources, including the use of over 120,000 bed days. The average tariff paid for these admissions ranged from £1,629 to £2,919 and the total reimbursement paid in England was £60 million [3].
Manual Chest Wall Physiotherapy (MCWP) is the standard treatment for managing airway clearance; MCWP involves techniques aimed at clearing the lungs by percussion, vibration, deep breathing, and huffing or coughing. The positioning of the patient during chest wall physiotherapy is important as posture can assist with secretion drainage. Community respiratory physiotherapy teams train carers in these techniques. A typical session takes about 30 minutes but can range from 10 to 60 minutes. Patients typically require 2 to 4 sessions a day but may need up to 6 sessions daily if they have a respiratory infection. As children grow older and into adulthood, maintaining the best position to support drainage can be very difficult for a carer providing the therapy on their own. This places a high burden on patients and carers who find it difficult to complete MCWP as frequently and effectively as recommended. This can result in insufficient airway clearance, recurrent respiratory infections, and increased health resource use.
The Vest™ system is an effective method to support airway clearance for patients and carers. It uses High-Frequency Chest Wall Oscillation (HFCWO) to loosen secretions in the chest by reducing their viscosity, which helps secretions move from smaller to larger airways where they can be cleared by coughing and suctioning [4, 5]. A treatment session usually takes 10 to 30 minutes. Studies have shown that this therapy can reduce hospital admissions, antibiotic use, and length of time spent in hospital [4]. Yuan et al. and Plioplys et al. [4, 5] both reported no adverse events associated with HFCWO in patients with cerebral palsy and neuromuscular disease, suggesting that the Vest™ is well tolerated and safe.
With over 30,000 people with complex neurological disorders living in the UK, it is important to know how the implementation of a novel airway clearance treatment will affect overall hospital admissions and bed days used. To date, a rigorous economic analysis of the cost-effectiveness of different airway clearance interventions has not been performed in the UK. The objective of this study is to evaluate the cost-effectiveness of the Vest™ System versus MCWP for airway clearance in patients with neuromuscular disease and cerebral palsy.
2. METHODS
2.1. Model Overview
A de novo decision-analytic Markov model was developed (Fig. 1). Costs and effects were estimated over 5- and 10-year time horizons. The model was used to simulate airway clearance in patients with cerebral palsy and other complex neurological disorders receiving one of two strategies: (1) the Vest™ (intervention) or (2) MCWP (comparator). Outcomes in the model were: total costs, total effectiveness (i.e. number of respiratory infections and hospital admissions), and incremental cost per Quality-Adjusted Life-Year (QALY) gained. The recommended discount rate in the UK (i.e. 3.5%) was used to discount costs and outcomes occurring after one year [6].
The Markov model comprises four health states with a cycle length of one month. The model structure is presented in Fig. (1). In each strategy, the patients may or may not experience a respiratory infection needing treatment or admission to hospital. The main input parameters in the model were: rate of respiratory infection, rate of hospitalisation, and antibiotic use for treatment of respiratory infection. In the model, patients were also at risk of death from any cause, or due to respiratory infection, during any cycle period. The cycle-specific risks of mortality were dependent on the cohort’s age. The risk of mortality was therefore increased with each cycle period. All-cause mortality rates were derived from general population mortality statistics reported in national life tables (Office for National Statistics) [7] and were adjusted to reflect the extra mortality associated with neuromuscular disease and cerebral palsy. Previous studies have shown that these people are at a higher risk of death compared to the general population with a standardized mortality ratio of 4 to 8.4 [8, 9]. Results from a UK-based study were also used to incorporate the mortality associated with respiratory infection [10]. All inputs used in the economic model, including information on clinical effectiveness, costs, and quality-of-life related to the different strategies are described below.
2.2. Model Inputs
2.2.1. Clinical Effectiveness
Table 1 provides an overview of the clinical inputs that were used in the model. Evidence has shown that patients are treated with HFCWO therapy as is provided by The Vest™, hospital admissions and length of stay in hospital decrease [4, 5]. A number of centres in England have initiated the use of HFCWO using the Vest™. Data collected by these services confirm the findings from the publications referenced above. One centre (Leicester) retrospectively collected data on NHS resource use for 8 severely disabled patients with cerebral palsy. After 12 months using the Vest™, there was a reduction in chest infections, antibiotic treatment, and hospital admissions as well as the length of stay when admission was required. These rates were converted to monthly probabilities using the following formula (p=1-exp(-rt)), where (p = probability of an event within one month) r = reported rate and t = time, and were used in the model in the base-case analysis.
Alternative values for the clinical inputs, which were used within the sensitivity analyses, were obtained from two different studies [4, 11]. The first study was a prospective, randomized, controlled trial performed by Yuan et al. [4]. In this study, 28 participants with neuromuscular disease or cerebral palsy were studied for a mean of 5 months. The second study was a cohort study comparing healthcare claims before and after initiation of HFCWO using the Vest™ [11]. In this study, data for 426 patients were analysed. Patients were in the database for an average of 16.9 months pre-intervention and 20 months post-intervention.
 |
Fig. (1). Model structure HFCWO: High Frequency Chest Wall Oscillation; MCWP: Manual Chest Wall Physiotherapy. |
| Parameters | Value | Distribution | LL | UL | Source |
|---|---|---|---|---|---|
| Clinical Input Parameters- Sourced from UK Based Study | |||||
| Probability of chest infection episodes per month-Pre VestTM system | 28.3% | Beta | 21.26% | 35.43% | UK-based study |
| Probability of admissions per patient per month -Pre VestTM system | 8.9% | Beta | 6.71% | 11.19% | UK-based study |
| Probability of rate of antibiotic treatments per month-Pre VestTM system | 18.0% | Beta | 13.47% | 22.45% | UK-based study |
| Average length of stay in hospital in each admission | 11.78 | Gamma | 5.89 | 17.67 | UK-based study |
| Probability of chest infection episodes per month-Post VestTM system | 3.1% | Beta | 2.31% | 3.85% | UK-based study |
| Probability of admissions per patient per month ->Post VestTM system | 4.1% | Beta | 3.06% | 5.10% | UK-based study |
| Probability of rate of antibiotic treatments per month-Post VestTMsystem | 16.2% | Beta | 12.17% | 20.29% | UK-based study |
| Average length of stay in hospital in each admission | 3.75 | Gamma | 1.88 | 5.63 | UK-based study |
| Clinical Input Parameters- Sourced from Yuan et al. 2010 | |||||
| % of participants requiring hospitalization/IV antibiotics at 5 months - MCWP | 6.4% | Beta | 4.84% | 8.06% | Yuan et al. 2010 [4] |
| % of participants requiring PO antibiotics at 5 months - MCWP | 11.0% | Beta | 8.26% | 13.76% | Yuan et al. 2010 [4] |
| % of participants requiring hospitalization/IV antibiotics at 5 months - HFCWO (The VestTMsystem) | 0.0% | Beta | 0.00% | 0.00% | Yuan et al. 2010 [4] |
| % of participants requiring PO antibiotics at 5 months - HFCWO (The VestTM system) | 5.3% | Beta | 3.98% | 6.64% | Yuan et al. 2010 [4] |
| Clinical Input Parameters- Sourced from Lechtzin et al. 2016 | |||||
| Risk reduction in inpatient admissions | 20.2% | Fixed | NA | NA | Lechtzin et al. 2016 [11] |
| Risk reduction in inpatient bed days | 44.8% | Fixed | NA | NA | Lechtzin et al. 2016 [11] |
| Risk reduction inphysician office visits | 8.2% | Fixed | NA | NA | Lechtzin et al. 2016 [11] |
| Health Utility | |||||
| Health utility without respiratory infection | 0.40 | Beta | 0.3 | 0.5 | NG62 [15] |
| Health utility with respiratory infection | 0.21 | Beta | 0.16 | 0.27 | [16] |
| Mortality | |||||
| 30 days mortality associated with RTI | 45.9 | Log Normal | 36.8 | 55.2 | [15] |
| Standardized Mortality Rates (SMR) | 8.4 | Log Normal | 4.20 | 12.60 | [16, 17] |
2.2.2. Costs
Cost was estimated from the perspective of the UK NHS and Personal Social Services. The following costs were included: cost of the Vest™ device and the costs of treating respiratory complications, including hospital admission and medication. Unit costs for these resources were extracted from standard sources such as NHS reference costs [12], British National Formulary (BNF), Personal Social Services Research Unit (PSSRU) [13, 14] and manufacturer price lists (Table 3). Costs were measured in UK pound sterling (£) for the year 2018.
The list price of the Vest™is £6,995 per device. It was assumed that the air pulse generator has a lifespan of between 5 and 10 years. The inflatable garments come in various styles and sizes and need to be replaced every 4-6 years (garment prices range from £275 to £295). It was also assumed that people with complex neurological needs and their carers would usually have 1 or 2 training sessions at their home about how to use the Vest™ system and 1 or 2 follow-up calls to make sure treatment is being delivered correctly. It was assumed that the training and follow-up calls would be performed by the community respiratory physiotherapy team. Therefore, the estimated annual cost for treatment with the Vest™ would be between £988 (based on 10 years of use) and £1,766 (based on 5 years of use) per person. MCWP is usually performed by carers so there is no ongoing cost to the NHS. Costs associated with treatment for respiratory infections were applied to both treatment strategies.
2.2.3. Cost of Respiratory Complications
The cost of inpatient care for respiratory infection was obtained from NHS reference costs. The weighted averages of the non-elective long stay HRG currencies (DZ1 and DZ22) were used to estimate the cost of each respiratory-related hospitalisation. Results from the UK-based study of the Vest™ show that after using the Vest™, patients have fewer hospitalisations and reduced length of stay in hospital compared to the year before using the Vest (3.78 vs.11.3 days). The average length of stay in hospital for respiratory infection is 5.92 days, therefore the unit costs from NHS reference costs were adjusted to reflect the excess bed days used for patients in the comparator arm. For those patients who experience respiratory infection and are treated as an outpatient case, the cost for one visit to the GP and an antibiotic prescription was used (Table 2).
2.2.4. Utilities
The health utility scores included in the model were a measure of the quality of life among people with neurological disorders. Health utility values for the health state without respiratory infection (0.40) and the disutility associated with a respiratory infection (-0.186) were derived from NICE guidelines and previous studies [15-17] (Table 1).
2.3. Analysis
The cumulative estimates of cost and effectiveness were reported for the two strategies. We conducted deterministic and Probabilistic Sensitivity Analyses (PSA) to explore the uncertainty surrounding the results. The deterministic sensitivity analysis was used to test the impact of varying key parameter values used in the base case analysis. PSA was used to map the parameter uncertainty. To conduct the PSA, probabilistic distributions were assigned to each input in the model, and were used to randomly select new plausible values. Each new sampled value was used within the equations in the model, and the new results of the model were recorded. This process was repeated for 10,000 iterations to produce a distribution of results from the model.
3. RESULTS
Total and incremental costs and effectiveness, Incremental Cost-Effectiveness Ratios (ICERs), and the probability that each strategy is cost-effective at £20,000 and £30,000 Willingness-To-Pay (WTP) thresholds are presented in Table 3. Probabilistic results based on a Monte Carlo simulation with 10,000 iterations, in the form of cost-effectiveness scatterplots and Cost-Effectiveness Acceptability Curves (CEACs), are presented in Figs. (2 and 3).
Over a 5-year time horizon, airway clearance using the Vest™ is less costly (£14,176 versus £20,211) per patient, and more effective (total QALYs per patient over 5 years = 1.72 versus 1.65) than MCWP; it is, therefore, a dominant strategy. The estimated total costs per patient using a 10-year time horizon were £24,108 versus £34,632, respectively, for the Vest™ and MCWP strategies. Total estimated QALYs per patient over the 10-year time horizon were 3.23 versus 3.03 for the Vest™ and MCWP strategies, respectively.
The CEACs shown in Figs. (2 and 3) indicate that over a 5-year time horizon, the Vest™ strategy has a high probability (>98%) of being cost-effective at WTP thresholds £20,000 and £30,000. In addition, results from the PSA show that the Vest™ strategy has a high likelihood (100%) of being a cost-saving strategy. Although the cost of the initial intervention is greater in the Vest™ strategy, cost savings, due to a reduction in the number of respiratory complications, result in an overall cost saving of approximately £6 million per 1,000 patients with complex neurological disorders in the UK over a 5-year time horizon. The estimated total hospital admissions and bed days saved per 1,000 patients were 2,442 and 49,868, respectively (Table 4).
The results from the deterministic sensitivity analysis showed that when results from the study by Yuan et al. [4] or Lechtzin et al. [11] are used to inform the clinical input parameters, the findings remain the same (Table 3).
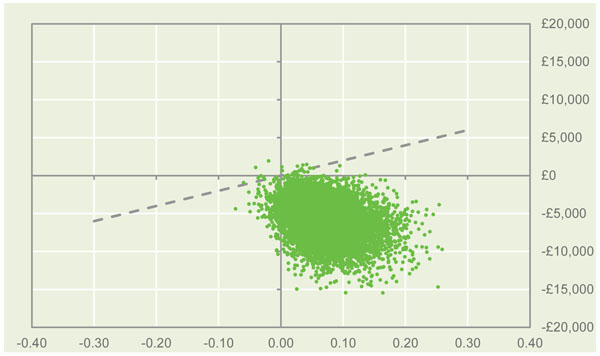 |
Fig. (2). Cost-effectiveness scatter plot at £20,000 WTP threshold. |
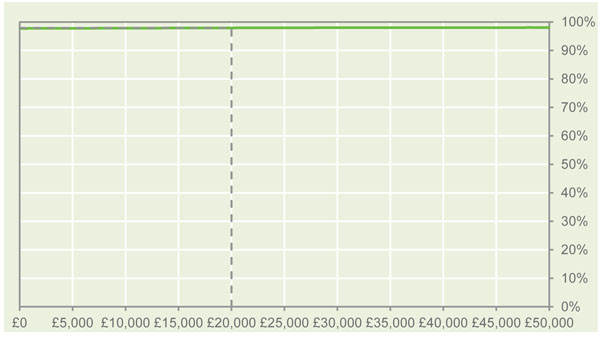 |
Fig. (3). Cost-effectiveness acceptability curve at £20,000 WTP threshold. |
| Resource Use | Value | Source |
|---|---|---|
| Cost of the Vest Equipment | £6,995 | Hill Rom company price list |
| Lifespan (yrs.) | 5-10 years | Assumption |
| Consumables | ||
| Wraps: Unit cost | £275 | Hill Rom company price list |
| Lifespan | 5 | Assumption |
| Vest: Unit cost | £295 | Hill Rom company price list |
| Lifespan | 5 | Assumption |
| Staff training | ||
| Band 7 PT: Unit cost (per session) | £87 | PSSRU [13] |
| Sessions | 1 | Assumption |
| Avg. patients trained in each session | 5 | Assumption |
| Patient training | ||
| Band 7 PT: Unit cost (per session) | £87 | PSSRU [13] |
| Hours Home visits, incl travel | 3 | Assumption |
| # home visits | 2 | Assumption |
| Hours phone follow-up | 1 | Assumption |
| # phone follow-up | 2 | Assumption |
| Cost of hospital admission | £2,444.26 | NHS reference cost 2017-18 (Non-elective long stay DZ1 and DZ22) |
| Cost per bed/day | £289.01 | NHS reference cost 2017-18 (Non-elective excess bed days DZ1 and DZ22) |
| Cost of medication | 1.37 | BNF [14] |
| Cost of GP visit | £37.40 | PSSRU [13] |
| Strategy | Average Cost Per Patient (£) | Incremental Cost (£) | Average QALYs Per Patient (£) | Incremental QALYs | ICER (£) (∆Cost/∆QALYs) | Probability Cost-Effective for Different WPT Thresholds (%) | |
|---|---|---|---|---|---|---|---|
| £20,000 | 30,000 | ||||||
| Base-case analysis- Informing the clinical effectiveness input parameters using results from the UK based study- 5 years’ Time horizon | |||||||
| MCWP | 20,211 | - | 1.65 | - | - | - | - |
| The Vest™ system | 14,176 | -6,035 | 1.72 | + 0.07 | Dominant | 98% | 99% |
| Base-case analysis- Informing the clinical effectiveness input parameters using results from the UK based study- 10 years’ Time horizon | |||||||
| MCWP | £34,632 | - | 3.03 | - | - | - | - |
| The Vest™ system | £24,108 | -10,524 | 3.23 | 0.19 | Dominant | 98% | 100% |
| Sensitivity analysis- Informing the clinical effectiveness input parameters using results from study by Yuan et al. 2010- 5 years’ Time horizon | |||||||
| MCWP | £14,835 | - | 1.77 | - | - | - | - |
| The VestTM system | ££9,108 | -£5,727 | 1.90 | 0.14 | Dominate | 100% | 100% |
| Sensitivity analysis- Informing the clinical effectiveness input parameters using results from study by Lechtzin et al. 2016- 5 years’ Time horizon | |||||||
| MCWP | £20,211 | - | 1.65 | - | - | - | - |
| The VestTM system | £17,618 | -£2,593 | 1.69 | 0.03 | Dominate | 100% | 100% |
| Health Resource Use | Pre-Vest™ | Post-Vest™ | Difference |
|---|---|---|---|
| Intervention cost per 1,000 patients | £230,580 | £7,820,998 | £7,590,418 |
| Total cost of hospital admissions per 1,000 patients | £19,487,088 | £6,016,681 | -£13,470,407 |
| Total cost of outpatient treatment per 1,000 patients | £493,269 | £338,594 | -£154,675 |
| Total cost per 1,000 patients | £20,210,937 | £14,176,272 | -£6,034,665 |
| Total number of hospital admissions per 1,000 patients | 5,071 | 2,630 | -2,442 |
| Total number of bed days per 1,000 patients | 59,729 | 9,861 | -49,868 |
| Total number of hospital admissions per patient | 5.07 | 2.63 | -2.4 |
| Total number of bed days per patient | 59.73 | 9.86 | -49.9 |
4. DISCUSSION
In the UK, respiratory illnesses result in significant economic costs for individual patients, as well as for the health service and society [18]. Patients suffering from neuromuscular disorders and cerebral palsy are at particular risk of experiencing respiratory problems because these conditions can result in a weakened diaphragm and respiratory muscles [19] leading to problems with airway clearance. There is currently a lack of published evidence available comparing emerging HFCWO techniques with alternative airway clearance methods in the UK and Europe for the treatment of respiratory problems in patients with complex neurological disorders [20]. The work presented here aims to add to the limited amount of cost-effectiveness literature available in this clinical area.
A decision-analytic Markov cohort model was developed to estimate the cost-effectiveness of introducing the Vest™ system, which utilises HFCWO, for the treatment of respiratory problems in patients with neuromuscular disease and cerebral palsy, compared to current MCWP methods. The analysis was carried out from a UK NHS perspective. Parameter values, including the time horizon and clinical inputs, were varied in sensitivity analyses to assess the impact that this would have on the model results.
Results indicate that the Vest™ system is a cost-effective strategy, with significant cost savings being made, primarily due to a reduction in required hospital admissions and a reduced length of stay for those patients who are admitted and therefore, lower associated costs. These findings remain consistent for all sensitivity analyses presented. Results from analysing health care utilization data from two large databases of insurance claims in the USA showed that the application of the VestTM system reduced the total medical costs per member per month by $1,949 [11].
There are, however, limitations to the analysis presented here. Firstly, the majority of the clinical effectiveness data used to populate the model were derived from two studies [4, 5], with additional data sourced from a UK-based study with only 8 patients. Despite the reliability of the identified studies, the low sample size in the UK-based data is a limitation of the analysis. However, we performed several sensitivity analyses to explore uncertainty including basing the analysis on clinical inputs from two other sources. In all of these sensitivity analyses the results were similar and the conclusion were generally the same. Secondly, cost and effectiveness data were only extrapolated over 5- and 10-year time horizons in this analysis. The lack of longer-term data is the key reason why the cost-effectiveness of each strategy could not be considered over the patient’s lifetime. Despite these limitations, we understand this to be the first cost-effectiveness study comparing the Vest™ system with current MCWP methods for the treatment of respiratory problems in patients with neuromuscular disease and cerebral palsy in a UK setting.
CONCLUSION
The Vest™ system has the potential to reduce the number of respiratory infections, respiratory-related hospitalisations experienced by patients with neuromuscular disease and cerebral palsy, which would result in cost savings to the NHS. Additionally, based on this analysis, the intervention could potentially improve patient outcomes, indicating that the Vest™ system has a high probability of being a cost-effective use of NHS resources.
LIST OF ABBREVIATIONS
AUTHORS' CONTRIBUTIONS
Mehdi Javanbakht was responsible for developing and populating the economic model and drafting the final version of the paper. All authors provided inputs for the model, read, and approved the final draft of the manuscript.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The authors declare that all of the data supporting the findings of this study are available within the article.
FUNDING
This report is independent research funded by Hill Rom Company.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.
REFERENCES
| [1] | Hull J, Aniapravan R, Chan E, et al. British Thoracic Society guideline for respiratory management of children with neuromuscular weakness. Thorax 2012; 67(Suppl. 1): i1-i40. |
| [2] | Seddon PC, Khan Y. Respiratory problems in children with neurological impairment. Arch Dis Child 2003; 88(1): 75-8. |
| [3] | NHS. National Schedule of Reference Costs 2016/17. London: Department of Health 2017.https://improvement.nhs.uk/resources/ reference-costs/ |
| [4] | Yuan N, Kane P, Shelton K, Matel J, Becker BC, Moss RB. Safety, tolerability, and efficacy of high-frequency chest wall oscillation in pediatric patients with cerebral palsy and neuromuscular diseases: An exploratory randomized controlled trial. J Child Neurol 2010; 25(7): 815-21. |
| [5] | Plioplys AV, Lewis S, Kasnicka I. Pulmonary vest therapy in pediatric long-term care. J Am Med Dir Assoc 2002; 3(5): 318-21. |
| [6] | Excellence NIHCaC. Guide to the methods of technology appraisal 2013 NICE 2013.https://www.nice.org.uk/process/pmg9/resources/ guide-to-the-methods-of-technology-appraisal-2013-pdf-2007975843781 |
| [7] | Office for National Statistics (ONS). Interim Life Tables. Available from: https://www.ons.gov.uk/peoplepopulationandcommunity/births deathsandmarriages/lifeexpectancies/bulletins/nationallifetablesunitedkingdom/previousReleases |
| [8] | Ray S, Chatterjee S, Ruiz-Lozano P. Pediatric cerebral palsy life expectancy: Has survival improved over time? Pediatr Therapeut 2013; 3: 1. |
| [9] | Strauss D, Cable W, Shavelle R. Causes of excess mortality in cerebral palsy. Dev Med Child Neurol 1999; 41(9): 580-5. |
| [10] | Myles PR, Hubbard RB, Gibson JE, Pogson Z, Smith CJ, McKeever TM. Pneumonia mortality in a UK general practice population cohort. Eur J Public Health 2009; 19(5): 521-6. |
| [11] | Lechtzin N, Wolfe LF, Frick KD. Theimpact of high-frequency chest wall oscillation on healthcare use in patients with neuromuscular diseases. Ann Am Thorac Soc 2016; 13(6): 904-9. |
| [12] | Department of Health. Reference Costs 2015-16 2016. |
| [13] | Curtis L, Burns A. Unit Costs of Health and Social Care 2017, Personal Social Services Research Unit 2017.https://www.pssru.ac.uk/project-pages/ unit-costs/unit-costs-2017/ |
| [14] | British National Formulary. BNF Online https://www.medicines complete.com/mc/?utm_source=bnforg&utm_medium=homepage&utm_campaign=medicinescomplete |
| [15] | Cerebral palsy in under 25s: Assessment and management 2017.https://www.nice.org.uk/guidance/ng62/evidence |
| [16] | Hunter R. Cost-effectiveness of point-of-care C-reactive protein tests for respiratory tract infection in primary care in England. Adv Ther 2015; 32(1): 69-85. |
| [17] | Oppong R, Jit M, Smith RD, et al. Cost-effectiveness of point-of-care C-reactive protein testing to inform antibiotic prescribing decisions. Br J Gen Pract 2013; 63(612): e465-71. |
| [18] | Trueman D, Woodcock F, Hancock E. 2017. Estimating the economic burden of respiratory illness in the UK. Retrieved from https://www.blf.org.uk/what-we-do/our-research/economic-burden |
| [19] | Gilchrist JM. Overview of neuromuscular disorders affecting respiratory function. Semin Respir Crit Care Med 2002; 23(3): 191-200. |
| [20] | Bradley JM. High frequency chest wall oscillation in cystic fibrosis. Thorax 2010; 65(3): 189-90. |




